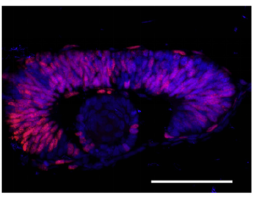
 Fig.1 Cdk1 regulates S phase initiation and/or DNA repair by phosphorylating Top2a at the S1213 site.
Fig.1 Cdk1 regulates S phase initiation and/or DNA repair by phosphorylating Top2a at the S1213 site.
Research Highlights
- Top2a is a potential downstream target, and cyclin A2 and cyclin B1 act as partners of Cdk1 in cell cycle regulation.
- The cyclin A2 depleted embryo phenotype replicates the cdk1-/- mutant, and the overexpression of Top2aS1213D rescued the S-phase defect.
- Cdk1 interacts with cyclin A2 to regulate S phase entry in part through Top2a phosphorylation, and interacts with cyclin B1 to regulate M phase progression.
The researchers found that the cyclin regulatory factors described in this study provide useful information for studying other zebrafish cyclins. The target of TOP2A, published in "Journal of Genetics and Genomics", may provide new opportunities for research areas such as Signal Pathway and Cell Biology. The scientist’s research is committed to facilitating cell proliferation and early development research.
The genetic changes of human TOP2A are related to cancer susceptibility. TOP2A separates chromosomes and is therefore necessary for many aspects of cell division (including DNA replication, chromosome condensation, and separation). Mammalian embryo development also requires TOP2A, because mouse Top2a knockout can cause embryo death as early as the 4-8 cell stage. Early studies concluded that maternal top2a enables zebrafish to develop before neutral transformation (MZT), while zebrafish top2a and top2b have no functional redundancy during development after activation of the zygotic genome. The previous article shows that inhibition or loss of Top2a leads to obvious cell cycle defects in the early embryonic cell cycle and later somatic cell cycle of zebrafish and Xenopus laevis. Among them, the blm mutant shows evidence of G2/M decatenation checkpoint activation. The latest research progress shows Top2a phosphorylation regulates zebrafish retinal development to S phase.
What is the role of cyclin A2 in zebrafish?
Cyclin A2/cyclin-dependent kinase 1-dependent phosphorylation of Top2a is required for S phase entry during retinal development in zebrafish. Cyclin-dependent kinase 1 (CDK1) plays an essential role in cell cycle regulation.
How are cyclin A2 and TOP2A related to Cdk1?
The author identified Top2a as a potential downstream target and cyclin A2 and cyclin B1 as partners of Cdk1 in cell cycle regulation via an in silico analysis. While depletion of either cyclin A2 or Top2a led to the decreased S phase entry in zebrafish retinal cells, the depletion of cyclin B1 led to M phase arrest.
What is the phosphorylation level of TOP2A mutants?
Top2a-/- mutants phenocopied cdk1-knockout ( cdk1-/-) mutants, and the phosphorylation level of serine 1213 (S1213) in Top2a was reduced in cdk1-/- mutants.
In this study, the research team uses the zebrafish model and addresses this knowledge gap by characterizing Cdk1 depleted embryos and identifying downstream factors. Finding that Cdk1 is necessary for retinal development because it regulates S phase entry and M phase progression. By analyzing the Cdk1 substrate and gene database necessary for retinal development, scientists verified that Top2a is a potential downstream factor of Cdk1.
The above conclusion is strongly supported by the following evidence. First, individual consumption of Cdk1, cyclin A2, and Top2a in zebrafish leads to similar phenotypes, including microphthalmia, decreased S-phase entry, and increased DNA damage in the retina. Consistent with these observations, it has been found that individual consumption of mouse Cdk1, cyclin A2, or Top2a can cause embryonic death at a very early stage. These findings indicate that the function of the cyclin A2-Cdk1-Top2a axis in cell cycle regulation is conserved in vertebrates. Secondly, in the zebrafish retina depleted of Cdk1- and cyclin A2, the phosphorylation of Top2a at S1213 was reduced, and the overexpression of TOP2A S1213D partially rescued cdk1-/retinal cell microphthalmia and S-phase entry defect-and ccna2-/- embryos. Third, the author's experimental data show that in S phase, cyclin A2 and Cdk1 interact in vitro and in vivo.
This article introduces that cyclin A2-Cdk1-Top2a plays an important role in regulating S phase, and the cyclin A2-Cdk1-Top2a axis may also play a role in other cell cycle stages, such as chromosome segregation during mitosis. Top2a is famous for its function in chromosome segregation because the consumption or inhibition of Top2a can cause chromosomes to lag or bridge after mitosis. This requires continuous exploration and learning by scientists, and this document provides excellent literature material for future research.
The work has been published in the Journal of Genetics and Genomics.
Story Source: Jin, M., Li, J., Hu, R., Xu, B., Huang, G., Huang, W., ... & Cao, Y. (2021). Cyclin A2/cyclin-dependent kinase 1-dependent phosphorylation of Top2a is required for S phase entry during retinal development in zebrafish. Journal of Genetics and Genomics, 48(1), 63-74.
More Information: Hossain, M. S., Kurokawa, K., Akimitsu, N., & Sekimizu, K. (2004). DNA topoisomerase II is required for the G0‐to‐S phase transition in Drosophila Schneider cells, but not in yeast. Genes to Cells, 9(10), 905-917.
Creative Biolabs is committed to providing our customers with high-quality products for modelorg antibody research. Currently, we provide a complete list of high-quality antibodies targeting Top2a. Please browse our Top2a product catalog backed by our guarantee.